Liposomal Vitamin D benefits, need more data
Liposomal vitamin D: promising delivery, thin evidence
Liposomal vitamin D encapsulation does appear to improve absorption rate and short-term bioavailability by roughly 4–6× compared to standard oil-based formulations, but this advantage is dose-dependent, most pronounced in deficient individuals, and supported by only a handful of small, mostly industry-funded studies. No long-term randomized trials have demonstrated that liposomal delivery improves clinical outcomes like fracture prevention or immune function. The strongest clinical case for liposomal vitamin D exists in populations with fat malabsorption — pancreatic-insufficient cystic fibrosis patients, post-bariatric surgery patients, and those with cholestatic liver disease — where standard absorption pathways are compromised. For healthy adults taking adequate doses with meals containing fat, the incremental benefit over a well-formulated oil-based softgel remains uncertain and likely marginal.
The clinical evidence is real but narrow
The most rigorous pharmacokinetic data comes from a crossover trial of 18 healthy volunteers by Dałek et al. (2022, Nanomedicine), which gave subjects a single 10,000 IU dose of vitamin D3 in either liposomal or oily formulation with a 3-week washout between crossover. The liposomal formulation produced absorption rates of 4.01 × 10⁻⁴ per minute versus 7.03 × 10⁻⁵ per minute for the oily form — approximately 6× faster. This effect was most pronounced in participants who were severely vitamin D deficient at baseline. Żurek et al. (2023, Pharmaceutics) applied pharmacokinetic modeling to the same dataset and calculated that the liposomal formulation achieved roughly 4–5× greater AUC over a 30-day modeled period.
A separate four-arm double-blind RCT by Solnier et al. (2024, Nutrients) tested LipoMicel® (a micellar delivery system, technically distinct from classical liposomes) at 1,000 and 2,500 IU daily for 30 days in 35 healthy adults. At 1,000 IU, the micellar form produced an incremental AUC roughly 6× higher than the standard softgel (992 vs. 177 nmol·day/L, p<0.05). Critically, however, no significant difference emerged at the 2,500 IU dose. The authors also noted that micellar vitamin D at 1,000 IU achieved serum 25(OH)D levels comparable to standard vitamin D at 2,500 IU, suggesting a potential 2.5× dose reduction with enhanced delivery — but only at lower doses.
An industry-funded study by ActiNovo tested liposomal D3+K2 against a standard tablet in 20 volunteers and claimed 12.84× greater bioavailability. This extreme figure warrants skepticism: it measured only a 6-hour acute window and compared a liquid liposomal to a dry tablet — not a fair head-to-head of delivery technologies.
In the one malabsorption population studied rigorously, the picture is mixed. Nowak et al. (2022, Journal of Clinical Medicine) conducted a multicenter RCT in 100 pancreatic-insufficient cystic fibrosis patients, comparing a liposomal ADEK+K2 formulation to standard vitamins over 3 months. The liposomal group achieved significantly higher 25(OH)D levels (43.2 vs. 32.7 ng/mL, p<0.001) and a change of +9.7 vs. +2.0 ng/mL (p=0.004). Yet an earlier three-arm trial by the same group (Nowak et al. 2021, Nutrients, n=75 CF patients) found that liposomal vitamin D did not significantly outperform MCT oil for vitamin D specifically — while cyclodextrin-encapsulated vitamin D did. The discrepant results likely reflect differences in formulation quality and study design, underscoring that not all liposomal products perform equally.
How liposomes change the absorption game
Standard vitamin D3 absorption is a multi-step process that depends heavily on functioning bile secretion, pancreatic lipase activity, intact intestinal mucosa, and adequate dietary fat. Cholecalciferol must be emulsified by bile salts into mixed micelles (~3–10 nm), diffuse through the intestinal mucus layer, enter enterocytes, get incorporated into chylomicrons, and drain into intestinal lymphatics. This entire chain is vulnerable to disruption at every step.
Liposomal encapsulation alters this pathway in several important ways. First, liposomes arrive pre-formed at ~100–200 nm — small enough to penetrate the intestinal mucus barrier (pore size ~300–500 nm) without requiring bile-mediated emulsification. Molecular dynamics simulations confirm that vitamin D3 (logP >7) embeds firmly in the phospholipid bilayer and cannot spontaneously transfer between lipid structures across aqueous barriers — its bioavailability is therefore entirely determined by the fate of the liposome itself (Dałek et al., 2022).
Second, the phospholipid composition of liposomes mimics biological cell membranes, enabling direct fusion with enterocyte plasma membranes and cellular uptake through clathrin-mediated endocytosis, caveolae-mediated endocytosis, or macropinocytosis. This reduces dependence on the transporter proteins and bile-salt-mediated passive diffusion that govern standard vitamin D uptake. Once inside enterocytes, liposomal vitamin D is incorporated into chylomicrons and enters the lymphatic system — the same route used by conventionally absorbed vitamin D, but arrived at more efficiently.
A common misconception is that liposomal delivery "bypasses first-pass metabolism." While lymphatic transport does indeed bypass the portal vein and liver, vitamin D actually requires hepatic 25-hydroxylation by CYP2R1 to form its major circulating metabolite, calcidiol. The real advantage is not avoidance of hepatic processing but rather more complete and rapid delivery to the liver via chylomicron remnants — reducing losses that occur during the conventional multi-step absorption process.
Bile salts in the intestine still interact with liposomes, further reducing their size by converting them into even smaller mixed micelles. This means liposomal vitamin D does not entirely bypass bile-dependent pathways; rather, it reduces the criticality of bile by providing a pre-formed structure that can be absorbed even when bile availability is limited.
Malabsorption populations stand to gain the most
The theoretical case for liposomal vitamin D is strongest where the standard absorption cascade breaks down.
Cholestatic liver disease and bile acid deficiency may represent the single strongest indication. Standard vitamin D absorption is fundamentally bile-dependent. Patients with primary biliary cholangitis, biliary atresia, or bile acid synthesis disorders can develop vitamin D-deficiency rickets refractory to mega-doses of oral vitamin D. Liposomal delivery provides pre-formed particles absorbable through membrane fusion and endocytosis without requiring bile-mediated emulsification — a genuine mechanistic bypass.
Post-bariatric surgery patients, especially after Roux-en-Y gastric bypass or biliopancreatic diversion, lose the duodenum and proximal jejunum — primary vitamin D absorption sites. Intestinal fractional calcium absorption can drop from ~33% to ~7% within 6 months of surgery. Despite this strong rationale, no published RCTs have directly compared liposomal to standard vitamin D in bariatric patients.
Cystic fibrosis with pancreatic insufficiency has the best direct evidence (the Nowak 2022 RCT), showing liposomal formulations circumvent the need for lipase-mediated fat digestion. Crohn's disease patients, particularly those with ileal involvement causing bile acid malabsorption, and celiac disease patients with active villous atrophy would similarly benefit from a delivery system that reduces dependence on intact mucosal surface area and bile function.
For elderly populations with age-related declines in bile secretion and pancreatic function, and for people on very low-fat diets (where co-ingested fat is insufficient to trigger adequate bile release), liposomal delivery eliminates the requirement for concurrent dietary fat — potentially improving both absorption and compliance.
How liposomal stacks up against other enhanced forms
Liposomal is not the only game in town. Several enhanced vitamin D delivery systems exist, each with distinct evidence profiles.
Oil-based softgels remain the gold standard for general use, with the most extensive evidence base and documented bioavailability of 55–99% in healthy adults. A systematic review confirmed that oil-soluble vehicles produce greater 25(OH)D increases than powder- or ethanol-based supplements. For healthy people taking supplements with fat-containing meals, these work well and cost pennies per day.
Water-soluble/micellar preparations like DEKAs and AquADEKs are specifically designed for malabsorption populations and have the strongest clinical evidence in CF — tested across 7 published studies. A French pediatric CF cohort showed that switching to DEKAs eliminated vitamin D deficiency over 12 months. These represent the current clinical standard of care for malabsorption.
Nanoemulsion vitamin D has shown 36% higher AUC and 43% higher Cmax versus softgels in a 24-person crossover study (Nandgaye et al.), though a 2025 IBD trial found similar 25(OH)D variation for buccal nanoemulsion spray versus conventional supplementation — a somewhat disappointing result in the population that should benefit most.
Buccal/sublingual sprays showed a ~2× absorption advantage over capsules in healthy subjects and ~2.6× in malabsorption patients in a crossover study by Satia et al. (2015, Nutrition Journal), offering an interesting GI-bypass alternative.
25-hydroxyvitamin D3 (calcifediol) deserves special mention: because it is more water-soluble than cholecalciferol and bypasses the hepatic hydroxylation step, patients with fat malabsorption absorb it equally well as healthy adults — versus 64% lower AUC with standard D3 (Holick et al., 2021). This pre-hydroxylated form addresses a fundamentally different bottleneck.
A notable animal study complicates the picture: in rats, microencapsulated vitamin D3 was absorbed 25% more than oil-based, while micellar vitamin D was absorbed only ~65% as well — the worst performer (Maurya et al., 2019, Medicina). This divergence from some human data highlights that formulation specifics matter enormously.
Real downsides temper the enthusiasm
Cost is substantial. Standard vitamin D3 softgels (5,000 IU) cost roughly $0.02–0.05 per day. Premium liquid liposomal products from brands like Quicksilver Scientific or Cymbiotika run $0.50–2.00 per day — a 10–40× premium. Liposomal softgels split the difference at roughly 2–3× the cost of standard softgels.
Quality control is arguably the most serious concern. A March 2025 analytical report on 21 top-selling Amazon NAD+ supplements found that among 11 products labeled "liposomal," 8 contained no detectable active ingredient, and the remaining 3 showed no functional liposomal structures despite containing sunflower lecithin. While this study examined NAD+ rather than vitamin D, it reveals systemic issues in the liposomal supplement market. There are no regulatory requirements for supplement manufacturers to demonstrate actual liposome formation, encapsulation efficiency, or particle size characterization. The term "liposomal" can be used without verification.
Stability requires careful handling. While liposomal encapsulation actually protects vitamin D3 from photodegradation better than free D3 (only ~5% loss vs. 48% after 9 days of light exposure), liquid formulations are sensitive to temperature. At 40°C, liposomal vitamin D showed ~8–9% decline after 9 days. Refrigeration is recommended. After opening, shelf life varies dramatically by product — from 11 days to over 22 months.
The dose-dependent absorption ceiling observed in the Solnier 2024 study is clinically important: the advantage over standard formulations disappeared at 2,500 IU. This suggests that at therapeutic doses used for deficiency correction (often 5,000–50,000 IU), the incremental benefit of liposomal delivery may be minimal in healthy individuals. Conversely, this same finding raises toxicity concerns at the lower end — if 1,000 IU liposomal truly delivers the equivalent of 2,500 IU standard, consumers taking higher liposomal doses could inadvertently approach harmful levels.
No study longer than 3 months has been published. No trial has examined clinical endpoints — fractures, infections, cardiovascular events, mortality — rather than surrogate serum markers.
Co-encapsulated K2 and magnesium add convenience, not proven synergy
Vitamin D3 and K2 (MK-7) are frequently co-encapsulated in liposomal products, based on their well-established complementary roles in calcium metabolism: D3 enhances intestinal calcium absorption by 30–40%, while K2 activates osteocalcin and matrix Gla-protein to direct calcium into bones and away from arteries. Both are fat-soluble and naturally suited to co-loading in phospholipid bilayers.
The CF RCT by Nowak et al. (2022) used a liposomal formulation containing both D3 and K2-MK7 and showed superior vitamin D delivery alongside improved vitamin K-dependent carboxylation of osteocalcin. However, the study design cannot separate the contribution of liposomal delivery from the addition of K2 itself, since the control group did not receive K2. The ActiNovo study found liposomal K2 was 37× more bioavailable than the standard tablet form — suggesting K2 benefits substantially from liposomal delivery — but this comes from an industry study comparing liquid to tablet.
No published study has directly compared co-encapsulated liposomal D3+K2 versus taking liposomal D3 and liposomal K2 separately. The theoretical advantages of co-encapsulation include simultaneous delivery to the same absorption site, simplified dosing, and combined protection from degradation — but these remain theoretical.
Magnesium co-encapsulation presents a different challenge. As a water-soluble mineral, magnesium does not naturally integrate into the lipid bilayer where D3 and K2 reside. Products marketing "liposomal D3+K2+magnesium" likely use magnesium in the aqueous liposomal core or as a separate non-encapsulated ingredient. Magnesium is an essential cofactor for the hepatic 25-hydroxylation and renal 1α-hydroxylation of vitamin D, and D3 supplementation can deplete magnesium stores, making co-supplementation reasonable. However, no clinical data specifically evaluates the benefit of co-encapsulated versus separately administered magnesium alongside liposomal D3.
Conclusion
Liposomal vitamin D represents a genuinely different delivery mechanism that reduces dependence on bile salts, dietary fat, and intact intestinal surface area for absorption. The pharmacokinetic advantage of 4–6× greater bioavailability is supported by small but legitimate crossover and RCT data — though this advantage shrinks at higher doses and in vitamin D-sufficient individuals.
The most compelling use case is in fat malabsorption conditions (CF with pancreatic insufficiency, cholestatic liver disease, post-bariatric surgery), where a single CF trial has shown meaningful clinical benefit.
For healthy adults, the case is weaker. Oil-based softgels taken with a fat-containing meal deliver vitamin D efficiently at a fraction of the cost. The liposomal supplement market is plagued by quality control problems, inflated marketing claims, and a regulatory vacuum that allows products to be labeled "liposomal" without verification of intact liposomal structures. Before choosing liposomal vitamin D, consumers should prioritize products from manufacturers that disclose particle size, encapsulation efficiency, and third-party testing data. The field urgently needs large, independent, long-term RCTs with clinical endpoints to determine whether the short-term bioavailability advantage translates into meaningful health outcomes.
Liposomal vs Standard Oral Vitamin D₃: Unique Benefits
Executive Summary
- Absorption & Bioavailability: Liposomal vitamin D₃ delivers D₃ more rapidly and efficiently than oil-based forms. In one PK trial of healthy adults (single 10,000 IU dose), liposomal D₃ produced ~5× higher serum 25(OH)D (calcidiol) C_max and 0–30 d AUC than a conventional oily solution. Notably, liposomal D₃ caused a significant 25(OH)D rise within hours, whereas the oil form showed essentially no early effect. The time to peak (≈14 days) and half-life (~43 days) were similar for both forms, indicating the advantage is in the onset and extent of absorption, not elimination.
- Clinical Efficacy: Human trials are limited and mixed. A 3-month RCT in cystic fibrosis (CF) patients showed a much greater 25(OH)D increase with liposomal multivitamins (+9.7 ng/mL) than with standard capsules (+2.0 ng/mL, p=0.004). By contrast, another CF trial found no significant benefit of liposomal D₃ over standard oil (mean Δ25(OH)D +3.7 vs +1.1 ng/mL, p=0.296). No large trials in healthy adults are reported. Thus clinical superiority is plausible but not conclusively proven.
- Safety & Tolerability: Liposomes (usually phospholipids) are generally safe as excipients. No serious adverse events have been attributed to liposomal vitamin D in trials. One CF trial noted a few gastrointestinal discomfort withdrawals on the liposomal regimen, but overall tolerability was similar to standard D₃. The safety profile otherwise matches that of vitamin D: excessive doses can raise calcium (risk of hypercalcemia) regardless of formulation.
- Dosing Differences: Formal dosing guidelines do not differ. Studies typically used equal IU doses for liposomal and standard D₃. Some manufacturers imply liposomal D₃ “works at lower doses”, but no consensus exists. In practice, one would start with similar IU amounts and monitor serum levels. If liposomal bioavailability is higher, equivalent serum targets might be reached with somewhat lower IU, but data on dose-adjustment are lacking.
- Stability & Formulation: Liposomal D₃ can improve solubility and stability of the vitamin. In vitro studies show liposomal encapsulation protects D₃ from oxidation and maintains content well (e.g. stable ≥28 days at 25 °C). In contrast, oil suspensions can separate and oxidize. However, many liposomal products require refrigeration (phospholipids can degrade at room temperature), which may be less convenient. Also, “liposomal” supplements vary greatly in quality; third-party seals (e.g. Specnova’s TruLiposome™) have been proposed but no official standard exists.
- Cost & Accessibility: Liposomal D₃ is a niche premium supplement. Retail prices are typically an order of magnitude higher per IU than standard D₃. For example, a 5000 IU liposomal D₃ capsule can cost ~$0.70 each, whereas a generic 5000 IU softgel is ~$0.07. Liposomal formulations are sold by specialty brands and usually in smaller doses or liquid form; they are not widely carried in basic multivitamins. Insurance or formularies generally do not cover them, and coupons/sales are limited.
- Regulatory Status & Claims: There is no separate regulatory category for liposomal nutrients. In the US and EU, liposomal D₃ products are regulated as dietary supplements. Companies cannot market them as “better drugs” unless approved. Indeed, FDA has issued warning letters to marketers using unsubstantiated health claims for “liposomal vitamin D₃” (e.g. immunity claims). Health claims must be structure/function (e.g. “supports bone health”) and substantiated by evidence. Some regulators scrutinize advanced formulations: for example, a liposomal D₃ product in Poland was initially classified as a medical food (FSMP) before being allowed as a normal supplement. In practice, labels often claim “enhanced absorption” without rigorous proof. No official health authority currently endorses specific claims for liposomal vitamin D.
- Real-World Evidence: Outside controlled trials, data are sparse. No large observational studies or post-market surveillance have examined long-term outcomes of liposomal vs standard D₃ supplementation. Consumer reports and testimonials (“I felt better on liposomal D₃”) exist but are anecdotal. Market analysts note growing interest in “nano” or “liposomal” supplements, but objective evidence (e.g. NHANES usage data) is not available. In summary, real-world performance is largely unmeasured and claims remain theoretical until further study.
Assumptions: This analysis assumes a general adult population with typical vitamin D dosing (a few thousand IU/day) and no specific conditions (unless noted). Unless otherwise specified, comparisons refer to cholecalciferol (D₃) in healthy individuals or common clinical settings.
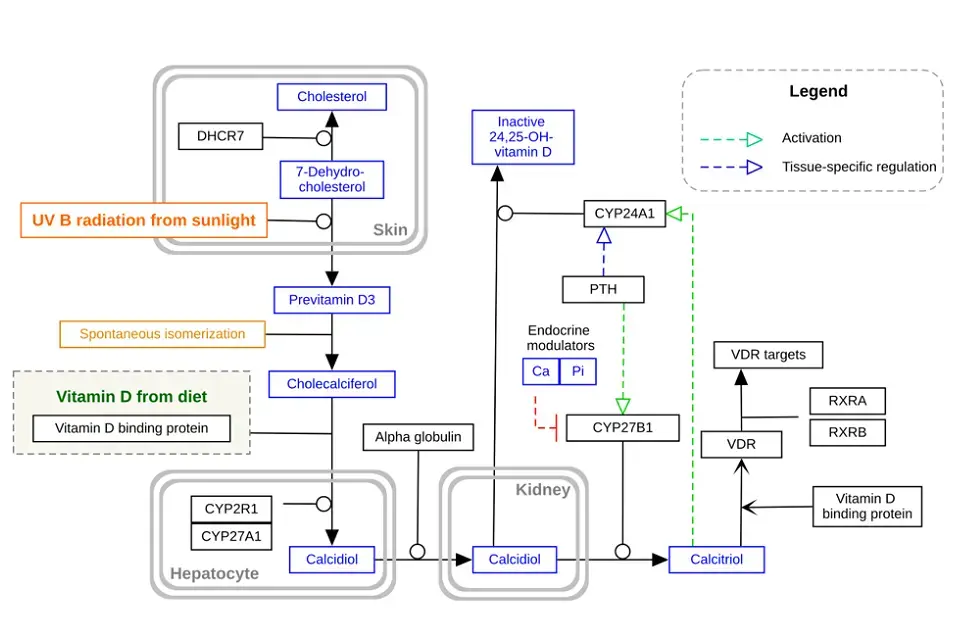
- Figure: Schematic of vitamin D₃ absorption and metabolism. Dietary cholecalciferol (D₃) is absorbed (requiring fat) and carried to the liver, where it is hydroxylated to 25-hydroxyvitamin D (25(OH)D). The kidney then produces the active 1,25-dihydroxyvitamin D. Liposomal formulations aim to improve intestinal uptake of D₃ via alternative pathways. (Adapted from Wikimedia Commons.)*
Pharmacokinetics (Absorption & Bioavailability)
Liposomal D₃ is designed to bypass some limits of fat absorption. Conventional vitamin D₃ in oil must form emulsified micelles aided by bile acids; these large heterogeneous oil droplets can coalesce or be excreted, limiting uptake. In contrast, liposomes (spherical phospholipid vesicles ~100–300 nm) can merge with enterocyte membranes or be transcytosed intact. A 2023 pharmacokinetic experiment confirmed this: after a single 10,000 IU dose, the liposomal formulation produced a rapid and large 25(OH)D rise in blood, whereas the oily form showed negligible early absorption. Quantitatively, the liposomal group’s 0–30 day area under the 25(OH)D concentration curve was ~8.5 ng·day/mL vs 1.69 ng·day/mL for oil (≈5× higher), and its Cmax was 0.335 vs 0.067 ng/mL. These results imply that more of the dose is absorbed with liposomes. Importantly, the Tmax (~14 days) and elimination half-life (~43 days) of 25(OH)D were identical between formulations, indicating liposomes affect the rate and extent of uptake but not the downstream metabolism. In practical terms, liposomal D₃ can raise serum 25(OH)D much faster and higher with the same dose.
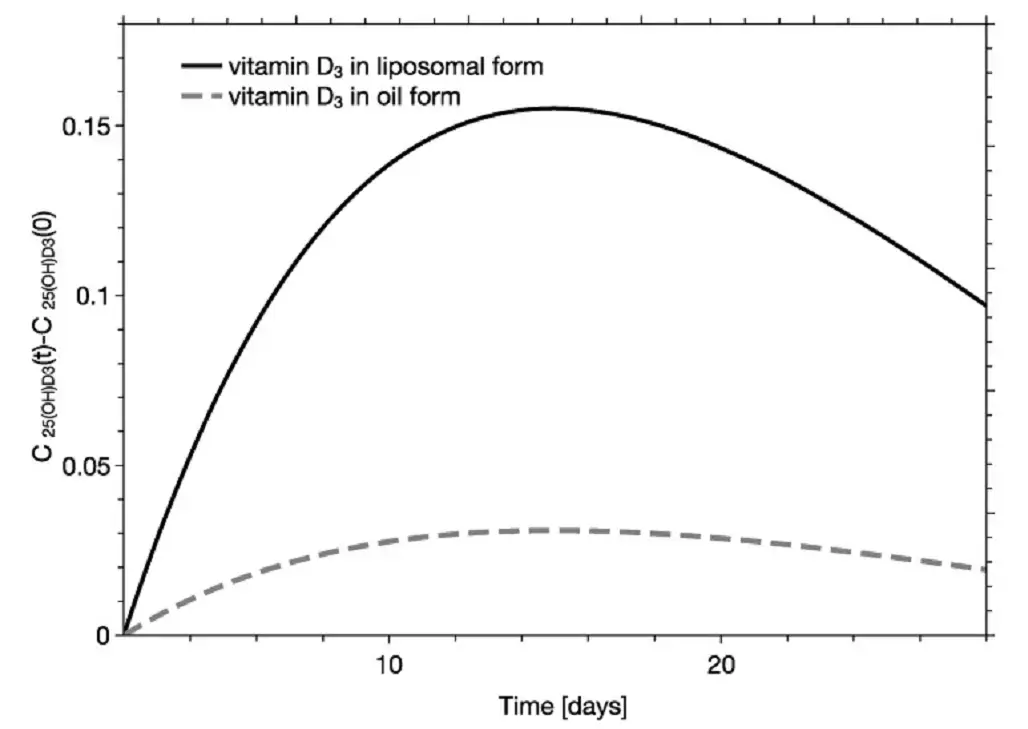
- Figure: Modeled serum 25(OH)D₃ response after a single oral dose of D₃. Black curve: liposomal formulation; gray: standard oil. The liposomal form achieves a much higher peak and area under curve (AUC) of 25(OH)D, reflecting superior absorption.*
Clinical Efficacy (25(OH)D Response & Dose-Response)
Few clinical trials directly compare liposomal vs standard D₃. The available evidence is limited and mixed. In one multicenter RCT in pancreatic-insufficient cystic fibrosis (CF) patients (n≈91), adults received either a liposomal multivitamin (including D₃) or a conventional vitamin supplement for 3 months. The liposomal group’s mean serum 25(OH)D rose by ≈9.7 ng/mL, whereas the standard group rose only 2.0 ng/mL (p≈0.004). This suggests a substantially better dose-response for the liposomal product in that setting. In contrast, another CF trial (pediatric/adolescent, n=67) found no significant difference between liposomal D₃ and a standard oil-based D₃ after 90 days. Both groups started with moderate deficiency; at 3 months 25(OH)D reached ~34 ng/mL (liposomal) vs ~31 ng/mL (oil), a non-significant gap. In that study a cyclodextrin-based D₃ increased levels more than either liposomal or oil.
No randomized trials in healthy adults or other populations are published to our knowledge. Thus, while the pharmacokinetic modeling strongly predicts higher efficacy of liposomal D₃, real-world serum 25(OH)D outcomes are not yet conclusively proven except in select trials. Meta-analyses or large RCTs are lacking; direct comparisons tend to be small and situation-specific. Overall, liposomal D₃ may achieve higher vitamin D levels at a given dose, but evidence to guide dosing adjustments or to confirm clinical benefit (e.g. bone health outcomes) remains limited. Dose-response curves beyond 25(OH)D levels (e.g. functional outcomes) have not been established.
Safety and Tolerability
Liposomal D₃ carries the same intrinsic safety concerns as regular vitamin D₃ (hypercalcemia, renal stone risk at very high intake). No unique toxicities of the liposomal delivery itself have been identified. In the multi-nutrient PK trial (healthy adults), liposomal preparations were well-tolerated with no serious adverse events or lab abnormalities. Similarly, the CF supplement trials reported no major safety signals. One CF study noted that 3 participants on liposomal vitamins withdrew due to gastric discomfort, whereas none withdrew in the standard-vitamin arm. This hints at occasional GI upset (possibly from the formulation or excipients) but not a systemic safety issue. An animal study (mice with tumors) found that liposomal D₃ delayed tumor growth and did not cause hypercalcemia, whereas high-dose free D₃ often does.
Phospholipid carriers are generally recognized as safe (GRAS), and liposomal products for other nutrients (e.g. vitamin C, glutathione) have similar tolerability. We note that hypersensitivity to soy/egg lecithin (if used) is a theoretical issue. In practice, no regulatory body has flagged liposomal D₃ as unsafe. Until high-dose trials are done, one should monitor calcium like with any vitamin D therapy. In summary, tolerability of liposomal D₃ is comparable to standard D₃, with only minor GI complaints reported sporadically.
Dosing Considerations
No formal guidelines specify different dosing for liposomal D₃. Manufacturers often suggest that liposomal forms may be used at lower IU for the same effect, but this is not evidence-based. Clinical studies to date have simply given equal IU amounts of liposomal vs standard D₃. For example, the PK trial used 10,000 IU of each form. Clinicians should still follow usual dosing based on patient needs (deficiency, body weight, etc.), and verify response with 25(OH)D assays. If liposomal D₃ is presumed more bioavailable, one could hypothesize that a somewhat smaller dose might suffice; however, without direct studies on dosing equivalence, it is safest to treat the IU dose the same and adjust after measuring levels. No dose-ranging studies (e.g. whether 5000 IU liposomal = 8000 IU oil) have been published.
Stability and Formulation Issues
Liposomes can improve the chemical stability of vitamin D. In vitro studies show that nanoliposomal D₃ remains intact in simulated gastric and intestinal fluids better than oil suspensions. Specifically, two prototype liposomes retained ~2 mg/mL of D₃ and remained stable in water and GI conditions, whereas oil droplets can coalesce and oxidize. One study demonstrated that lyophilized liposomes containing vitamin D were chemically stable for at least 28 days at room temperature and dissolved rapidly in tests. In practice, many commercial liposomal D₃ products are packaged as liquids or gel capsules with opaque foil to protect against light, and often include antioxidants to prevent oxidation.
On the downside, liposomal formulations often require special handling. For example, the CF trial shipped liposomal vitamins with cold packs and instructed patients to refrigerate them, whereas oily softgels can be stored at room temperature. Leaking or separating can occur if liposomal emulsions freeze or are overheated. Packaging costs may be higher (multi-dose vials, amber bottles). Moreover, “liposomal” is sometimes used loosely: some products may contain nanoemulsions or micelles rather than true bilayer vesicles, and analytical verification (e.g. cryo-EM, size distribution) is often not disclosed. Thus, product quality (particle size uniformity, encapsulation efficiency) can vary. Consumers should look for reputable brands that specify manufacturing and ingredients (e.g. phosphatidylcholine content) and possibly third-party validation.
Cost and Accessibility
Liposomal vitamin D₃ is significantly more expensive per IU than standard D₃. For example, a 30-count bottle of 5000 IU liposomal D₃ (Mercola brand) costs about $21 (≈$0.70 per capsule), whereas a common 180-count bottle of 5000 IU regular D₃ is about $12 (≈$0.07 per capsule). Even accounting for potential higher absorption, the cost difference is large (often 5–10× higher per dose). Liquid liposomal D₃ preparations (drops or sprays) also command higher unit prices.
These formulations are sold primarily through supplement retailers and online shops; they are not widely covered by health insurance or found in pharmacies as routine prescriptions. In many countries they are available only as over-the-counter dietary supplements. The high price and lower availability mean that liposomal D₃ may not be accessible or practical for many patients, especially when cost-effectiveness is considered.
Regulatory Status and Labeling Claims
In the US, liposomal vitamin D₃ products are regulated as dietary supplements. They must comply with all dietary supplement labeling rules (21 CFR § 101.36 for nutrition info, etc.) but there is no special category for “liposomal” on labels. Structure/function claims (e.g. “supports bone health”) can be used if evidence-backed, but disease-prevention or treatment claims are prohibited unless approved as drugs. The FDA has specifically warned companies against making unauthorized claims about liposomal vitamin C and D products. For example, one warning letter cited social media ads stating “Liposomal Vitamin D3… may potentially provide some protection for vulnerable populations” (referring to COVID-19), which the FDA flagged as an unapproved drug claim. The FDA also cautions that statements of absorption percentages (e.g. “90% absorbed”) must be truthful and not misleading; such figures for liposomal supplements typically lack independent validation.
In Europe, similar rules apply: health claims on supplements must be EFSA-approved. There are no authorized health claims specific to liposomal delivery. Companies may label “liposomal vitamin D3” or “advanced absorption D3,” but claims like “superior absorption” are not EFSA- or FDA-authorized. Notably, in Poland a marketed “Liposomal Vitamin D 4000 IU” was initially deemed a food for special medical purposes (FSMP) rather than a regular supplement, due to uncertainty about its ingredient classification. Only after additional review was it allowed as a standard supplement. This illustrates that some regulators scrutinize novel formulations. Overall, regulatory agencies treat liposomal D₃ the same as any vitamin D supplement; label claims must follow general laws. Buyers should view “liposomal” branding cautiously and rely on proven endpoints (serum levels) rather than promotional language.
Real-World Evidence
We found no independent real-world studies (e.g. cohort studies, pharmacoepidemiology) comparing liposomal vs standard D₃ in routine use. Market research suggests growing consumer interest in “novel” supplements, but published usage data (NHANES or other surveys) do not break out liposomal vs other forms. Some companies cite internal testing or testimonials, but such data are not peer-reviewed. In practice, most clinicians and patients rely on serum 25(OH)D monitoring to judge response, regardless of formulation. Until more post-market research is done, any “real-world advantage” of liposomal D₃ remains unverified. In summary, real-world evidence is entirely lacking; current conclusions come from small trials and lab studies.
Summary of Comparative Studies
| Study (Year) | Design | Population | Dose & Duration | Formulations | Key Findings (25(OH)D change) |
|---|---|---|---|---|---|
| Lukawski et al. (2022) | PK crossover trial | Healthy adults (n=24, age 24–65) | Single 10,000 IU dose | Liposomal D₃ vs oily solution | Liposomal: 5× higher 25(OH)D AUC₀₋₃₀d (8.51 vs 1.69) and C_max (0.335 vs 0.067 ng/mL) than oil. Serum rise seen within hours with liposomal but not oil. |
| Nowak et al. (2021) | Double-blind RCT | CF patients w/ pancreatic insufficiency (n=67, age 12–30) | ~3000–5000 IU/day for 90 days | Liposomal D₃ vs MCT oil (vs cyclodextrin D₃) | Liposomal group Δ25(OH)D ≈ +3.7 ng/mL; MCT oil +1.1 ng/mL (p=0.296, ns). Cyclodextrin D₃ increased +9.1 ng/mL (significantly more than oil, p=0.012). |
| Orchowska et al. (2022) | RCT, multi-center | CF adults (n=91) | Standard CF vitamin regimen (~6 months) | Liposomal multivitamins (A,D,E,K) vs standard vitamins | Liposomal group: mean Δ25(OH)D ≈ +9.7 ng/mL (±13.4); Standard group: +2.0 ng/mL (±9.8), p=0.004. At end, 25(OH)D rose to ~43 vs 33 ng/mL. |
Each study above shows the population and key result for vitamin D status. In short, absorption-type studies (Lukawski) find much greater 25(OH)D increase with liposomes, whereas clinical trials show mixed outcomes (one large trial favored liposomes, another found no significant difference). All cite serum 25(OH)D as the endpoint.
📝 RTF version of the above has hyperlinked citations
Related in Vitamin D Life
- Topical Vitamin D (nanoemulsion cream etc)
- Vitamin D significantly improved by encapsulation
- Vitamin D: liposomal and nanoemulsion forms - Chat-GPT
- Vitamin D spray results in bigger response than drops
- Nanoemulsion Vitamin D is faster and better - many studies
- Sublingual Vitamin D sometimes is better than tablet
- Nanoemulsion Vitamin D may be a substantially better form
- Overview Women and Vitamin D
