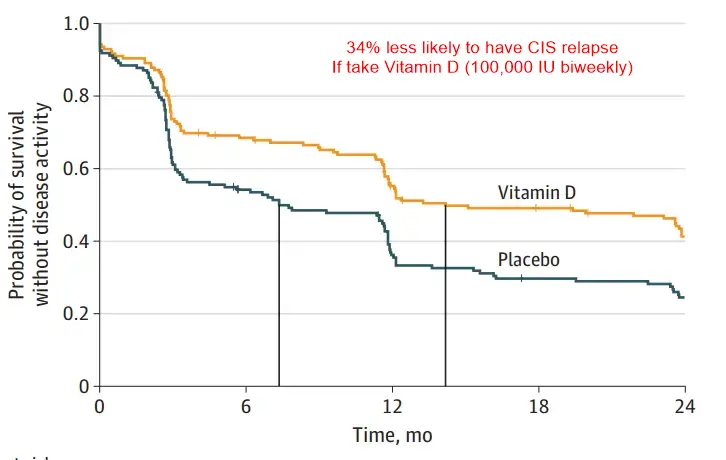
Clinically Isolated Syndrome (early MS) reduced 34% by bi-weekly 100,000 IU Vitamin D - RCT
High-Dose Vitamin D in Clinically Isolated Syndrome Typical of Multiple Sclerosis
The D-Lay MS Randomized Clinical TrialJAMA. 2025;333(16):1413-1422. doi:10.1001/jama.2025.1604 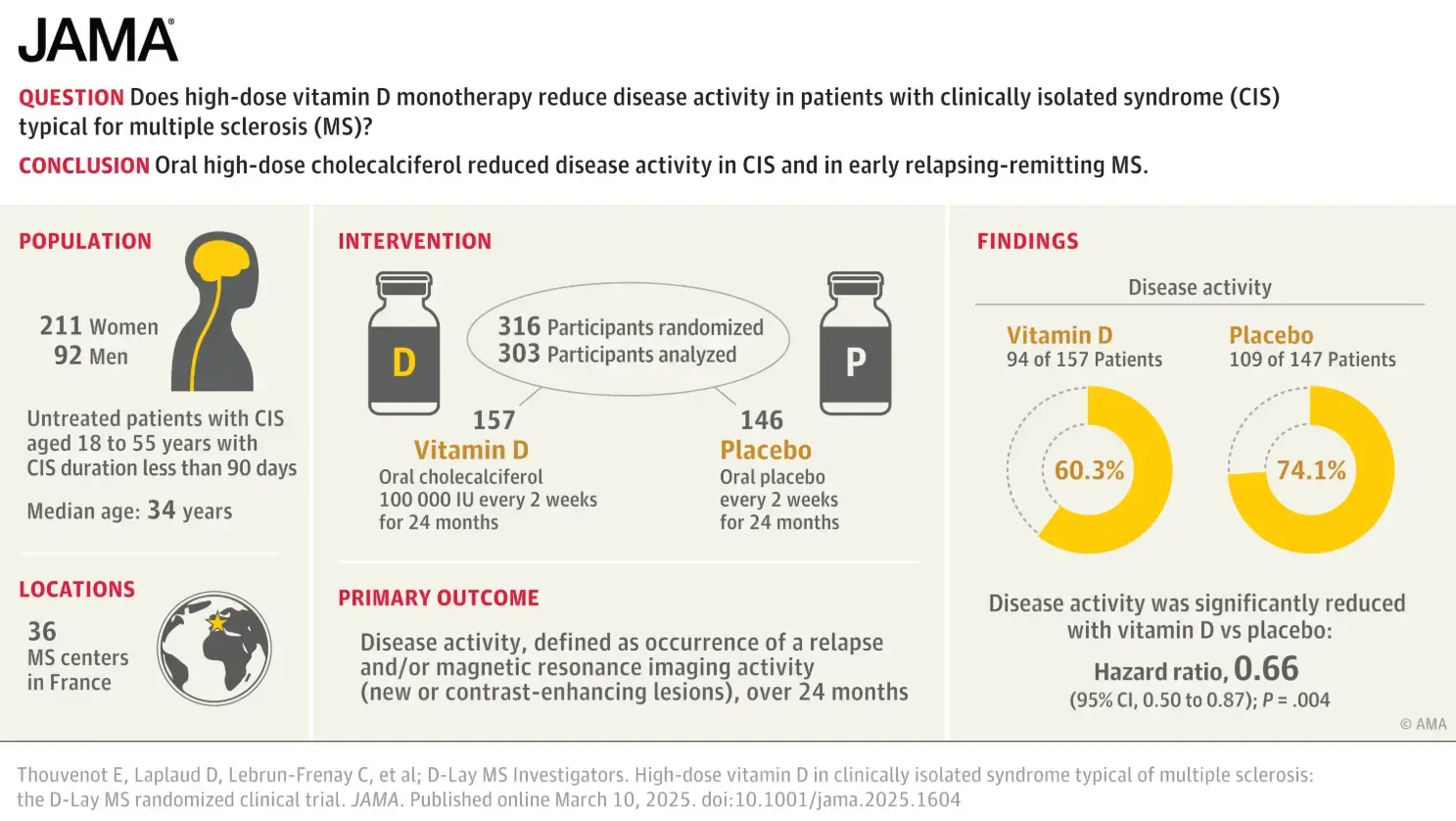
OBJECTIVE To evaluate the efficacy of high-dose cholecalciferol as monotherapy in reducing disease activity in patients with clinically isolated syndrome (CIS) typical for MS.
DESIGN, SETTING, AND PARTICIPANTS The D-Lay MS trial was a parallel, double-blind, randomized placebo-controlled clinical trial in 36 MS centers in France. Patients were enrolled from July 2013 to December 2020 (final follow-up on January 18, 2023). Untreated patients with CIS aged 18 to 55 years with CIS duration less than 90 days, serum vitamin D concentration less than 100 nmol/L, and diagnostic magnetic resonance imaging (MRI) meeting 2010 criteria for dissemination in space or 2 or more lesions and presence of oligoclonal bands were recruited.
INTERVENTION Patients were randomized 1:1 to receive oral cholecalciferol 100 000 IU (n = 163) or placebo (n = 153) every 2 weeks for 24 months.
MAIN OUTCOMES AND MEASURES The primary outcome measure was disease activity, defined as occurrence of a relapse and/or MRI activity (new and/or contrast-enhancing lesions) over 24 months of follow-up, also analyzed as separate secondary outcomes.
RESULTS Of the 316 participants enrolled and randomized (median [IQR] age, 34 [28-42] years; 70% women), the primary analysis included 303 patients (95.9%) who took at least 1 dose of the study drug and 288 (91.1%) ultimately completed the 24-month trial. Disease activity was observed in 94 patients (60.3%) in the vitamin D group and 109 patients (74.1%) in the placebo group (hazard ratio [HR], 0.66 [95% CI, 0.50-0.87]; P = .004), and median time to disease activity was longer in the vitamin D group (432 vs 224 days; log-rank P = .003). All 3 secondary MRI outcomes reported significant differences favoringthe vitamin D group vs the placebo group: MRI activity (89 patients [57.1%] vs 96 patients [65.3%]; HR, 0.71 [95% CI, 0.53-0.95];P = .02), new lesions (72 patients [46.2%] vs 87 patients [59.2%]; HR, 0.61 [95% CI, 0.44-0.84]; P = .003), and contrast-enhancing lesions (29 patients [18.6%] vs 50 patients [34.0%]; HR, 0.47 [95% CI, 0.30-0.75]; P = .001). All 10 secondary clinical outcomes showed no significant difference, including relapse, which occurred in 28 patients (17.9%) in the vitamin D group vs 32 (21.8%) in the placebo group (HR, 0.69 [95% CI, 0.42-1.16]; P = .16). Results were similar in a subset of 247 patients meeting updated 2017 diagnostic criteria for relapsing-remitting MS at treatment initiation. Severe adverse events occurred in 17 patients in the vitamin D group and 13 in the placebo group, none of which were related to cholecalciferol.
CONCLUSIONS AND RELEVANCE Oral cholecalciferol 100 000 IU every 2 weeks significantly reduced disease activity in CIS and early relapsing-remitting MS. These results warrant further investigation, including the potential role of pulse high-dose vitamin D as add-on therapy.
TRIAL REGISTRATION ClinicalTrials.gov Identifier: NCT01817166
Google AI summary of the 8-minute video
- (00:00) Despite extensive observational data linking low Vitamin D levels to poor outcomes in various diseases, past randomized trials have historically failed to prove that supplementation actually helps.
- (01:07) A recent randomized trial published in JAMA breaks this trend, demonstrating a clear, significant benefit of Vitamin D supplementation for multiple sclerosis (MS).
- (02:34) Unlike previous failed trials, this study targeted early intervention by enrolling 316 patients with "clinically isolated syndrome" (CIS), the earliest possible symptomatic presentation of MS.
- (03:08) The intervention group received aggressively high doses, taking 100,000 international units of oral Vitamin D3 every two weeks (roughly 100 times the dose of a standard over-the-counter pill).
- (05:29) The trial successfully reduced disease activity (symptom relapses or new MRI lesions), showing a 14% absolute risk reduction in the Vitamin D group (60%) compared to the placebo group (74%).
- (05:53) Treatment significantly delayed the progression of the disease, extending the median time to progression by approximately 7 months.
- (06:19) Subgroup analyses revealed that the Vitamin D supplements were most effective for patients who already had a baseline Vitamin D insufficiency and those with a milder initial disease presentation.
- (07:28) Despite the massive doses, the treatment proved to be highly safe; there were no cases of severe hypercalcemia or kidney failure reported in the Vitamin D group.
- (07:49) The success of this study suggests a blueprint for future Vitamin D research: successful trials may require catching the disease early and utilizing significantly higher doses of the vitamin.
Related in Vitamin D Life
- Multiple Sclerosis 2X-3X more likely if poor Vitamin D Receptor – Meta-analysis
- Treating Autoimmune Disease with the Coimbra Protocol - video Dec 2025
- Coimbra high-dose vitamin D protocol - many studies
- Comparing High-dose vitamin D therapies
All therapies use cofactors, some of which keep Calcium away from soft tissues
50,000 and 100,000 IU
- High-Doses (50,000 IU weekly, etc.) if at high risk of Vitamin D deficiency
- 50,000 IU and other high dose vitamin studies
- Multiple Sclerosis relapses cut in half by 100,000 IU of Vitamin D every 2 weeks– RCT 2019
- Delay onset of Multiple Sclerosis with 100,000 IU of vitamin D every two weeks – RCT 2020
- No multiple sclerosis relapses during pregnancy if 50,000 IU of Vitamin D weekly – RCT
- One pill every two weeks gives you all the vitamin D most adults need
