Lupus risk is reduced by diet - Omega-3, Vitamin D, Curcumin, etc - review of 105 studies
Dietary and Nutritional Factors in SLE Pathophysiology — Scoping Review - 2026
Citation: Jaya JN, Pudjihartono M, Pudjihartono N, Damara I, Nurkholis F, Zulhendri F. Advances in Nutrition (2026), journal pre-proof, doi:10.1016/j.advnut.2026.100644. Funded by the Efi Propolis Research Fund.
Method. PRISMA-guided scoping review of 139 studies (Scopus, PubMed, EBSCO; Jan 2012 – Jul 2023) covering observational, in vitro, animal, and clinical human studies on diet–SLE relationships. Search terms: "lupus AND ((food) OR (diet) OR (nutrition) OR (nutrient))." Studies were classified into thematic categories: macronutrients, ultra-processed foods/general dietary factors, alcohol, iodine, vitamin D, essential fatty acids, and natural products.
Key findings by category
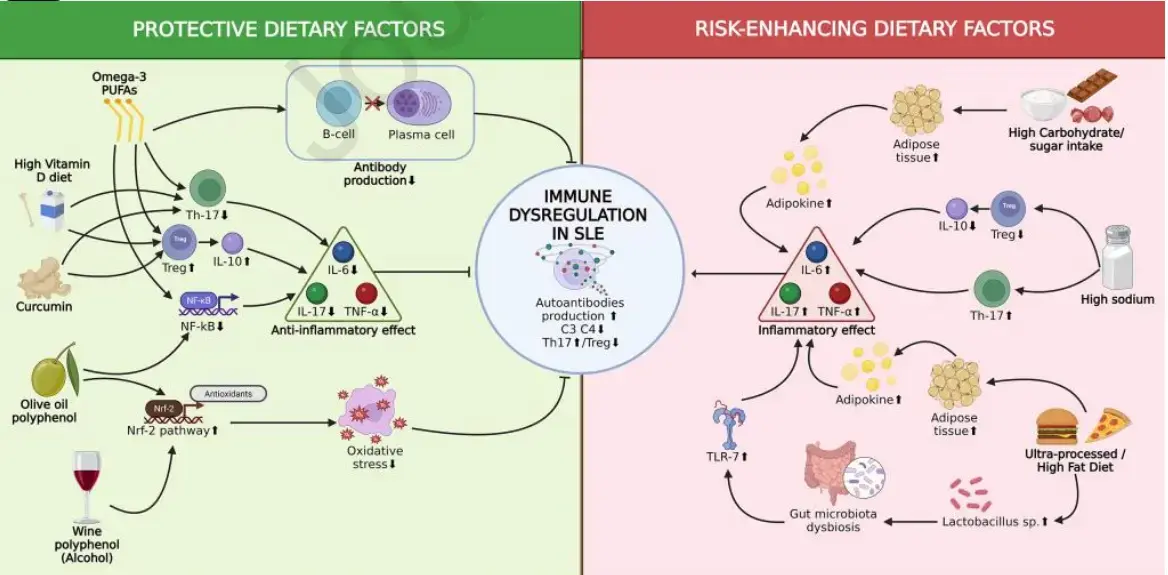
Macronutrients & general diet. Higher free-sugar intake and sugar-sweetened beverages tracked with greater disease activity, dyslipidemia, and increased SLE incidence (Correa-Rodríguez 2020; Castro-Webb 2021; Julià 2021). Higher MUFA and saturated fat intake was inversely associated with SLE risk in the Black Women's Health Study. A healthy lifestyle index in the Nurses' Health cohorts (185,962 women) was associated with roughly halved SLE incidence. Counter-evidence exists: Tedeschi (2020) and Barbhaiya (2021) found no significant association between AHEI-2010, aMed, DASH, or EDIP patterns and SLE risk. In mouse models, Western and high-fat diets worsen lupus via TLR7 signaling and microbiota shifts.
Essential fatty acids. The most consistent signal in the review. Omega-3 (EPA/DHA) supplementation in human RCTs (Arriens 2015: 2.25 g EPA + 2.25 g DHA × 6 mo; Borges 2017: 1080 mg EPA + 200 mg DHA × 12 wk) improved PGA, RAND SF-36, and CRP. Erythrocyte membrane PUFA studies showed higher omega-6:omega-3 ratios correlated with higher CRP and SLAQ scores. Murine work (cSiO2 and lupus-prone models) demonstrated DHA suppresses B/T-cell proliferation, follicular dendritic cell expansion, anti-dsDNA IgG, interferon-regulated gene expression, ectopic lymphoid tissue formation, and glomerulonephritis. Pestka 2014 showed omega-3-rich diets downregulate CD80, CTLA-4, IL-6, TNF-α, CCL-5, CXCR3 in kidney and spleen relative to omega-6/omega-9 diets.
Alcohol. Moderate intake (≥5 g/day, ~0.5 standard drinks) associated with reduced SLE incidence (HR 0.61) in the Nurses' Health cohorts (Barbhaiya 2017), with wine showing the strongest effect — pointing to polyphenols (resveratrol) rather than ethanol per se. Hahn 2020 found cumulative alcohol intake lowered circulating stem cell factor, an autoantibody-independent mechanism. The authors caveat the J-/U-shaped curve seen for RA, CVD, and all-cause mortality.
Sodium. High-sodium diets accelerate disease in MRL/lpr and NZBWF1 mice via the SGK1 → Th17 / Th1 axis, p38 MAPK-STAT1-mediated dendritic cell maturation, raised anti-dsDNA, and lowered C4. A clinical trial showed low-sodium intake reduced Th17, raised Tregs, and lowered IL-9 — effects reversed on resumption of normal sodium. Cross-sectional data linked higher sodium intake to elevated hsCRP and anti-dsDNA in SLE patients, plus gut permeability changes (Megamonas funiformis, plasma zonulin).
Vitamin D. Deficiency is consistently observed in SLE: Islam 2019 meta-analysis (34 case-control studies, 2,265 patients) confirmed inadequate 25(OH)D in SLE vs. controls; García-Carrasco 2017 found 126/137 SLE patients had insufficiency (<30 ng/mL) or deficiency (<10 ng/mL). Mechanisms covered include VDR expression in both innate and adaptive immune cells, macrophage conversion of 25(OH)D to calcitriol with cathelicidin induction, suppression of IL-6/TNF-α, regulation of Th1/Th17/Treg balance, and modulation of B-cell differentiation. Intervention evidence: Cutillas-Marco (CLE patients, 1400 IU cholecalciferol + 1250 mg Ca × 40 days, then maintenance × 1 year) showed disease severity improvement. Piantoni 2015 used an intensive cholecalciferol regimen (300,000 IU loading + 50,000 IU/month, ~850,000 IU/year) and observed CD4+CD45RA+CCR7− T-cell increases, CD8+CD28− decreases, and reduced IFN-γ/IL-4 in CD8+ cells. Franco 2017 meta-analysis: vitamin D supplementation reduces anti-dsDNA positivity. NZB/W F1 mouse data show correction of vitamin D delays proteinuria, reduces anti-dsDNA, increases IL-10 and regulatory CD4+ T cells, and attenuates the interferon-stimulated gene signature plus endothelial dysfunction.
Natural products. Olive oil phenolics (EVOO, VOO) showed immunomodulatory effects in murine SLE models. Curcumin (12.5–1000 mg/kg, pristane and MRL/lpr models) dose-dependently reduced Th1, Th2, Th17, Th17/Treg ratio, IL-6, TNF-α, ANA, BUN, proteinuria, and anti-dsDNA. Human curcumin RCTs in renal disease (including lupus nephritis) showed positive effects on inflammatory/oxidative markers but limited clinical impact beyond proteinuria. Other promising extracts: lactoferrin (hepatic fibrosis), Gentiana macrophylla (qin jiao, cardiac apoptosis), amaranth oil, isogarcinol, lycopene (lower mortality in retrospective cohort), caffeine (lower disease activity, cross-sectional). Notably, taurine supplementation worsened lupus in mice despite elevated serum taurine being observed in patients.
Synergy signal
A factorial RCT cited in the discussion found combined vitamin D + omega-3 supplementation reduced incident autoimmune disease, though the interaction term was non-significant — i.e., additive rather than true synergy on the available data. The authors flag combined-nutrient interventions as understudied.
Strengths & limitations (per the authors)
Strengths: spans the full translational continuum (in vitro → animal → observational → RCT) and explicitly identifies gaps between preclinical promise and clinical validation (curcumin is their flagship example). Limitations: no formal risk-of-bias assessment; no quantitative synthesis; thematic approach simplifies interactions; nutrients analyzed mostly in isolation; English-language only; 2012–2023 window; contradictory findings between studies; mouse-model SLE (pristane, cSiO2, NZB/W F1, MRL/lpr) doesn't fully recapitulate human heterogeneity, and animal-to-human dose translation remains uncertain. The authors call for large RCTs of multi-nutrient interventions.
Bottom line. The review concludes that diet — particularly omega-3 intake, vitamin D adequacy, low sodium, moderate alcohol/wine, and polyphenol-rich foods (olive oil, curcumin) — measurably modulates SLE risk and activity, and should be integrated as adjunct to pharmacotherapy. The strongest human evidence is for omega-3 supplementation and vitamin D repletion; curcumin and olive oil phenolics have strong preclinical but weak clinical evidence.
Related in Vitamin D Life
- Lupus summary of Vitamin D Life by Perplexity AI - Feb 2026
- Lupus
- Lupus: Vitamin D insufficiency nearly universal, should be an integral part of Lupus management
- Lupus treated again by daily Vitamin D - 4,000 IU or 8,000 IU (slightly better) - RCT
- Every aspect of Lupus is associated with low Vitamin D
- Lupus patients are 5 X more likely to have low vitamin D – many meta-analyses
- It is time to routinely give vitamin D to Lupus patients
Genes ( not mentioned by the study
- Lupus associated with another poor vitamin D gene (CYP27B1 – in addition to low Vitamin D and poor VDR)
- Lupus 3.8 X more likely if a poor Vitamin D Receptor
Omega-3
Curcumin
